Abstract
Background: Psychiatric surgery, including deep brain stimulation and stereotactic ablation, is an important treatment option in severe refractory psychiatric illness. Several large trials have demonstrated response rates of approximately 50%, underscoring the need to identify and select responders preoperatively. Recent advances in neuroimaging have brought this possibility into focus. We systematically reviewed the psychiatric surgery neuroimaging literature to assess the current state of evidence for preoperative imaging predictors of response.
Methods: We performed this study in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) and Meta-analysis of Observational Studies in Epidemiology (MOOSE) frameworks, and preregistered it using PROSPERO. We systematically searched the Medline, Embase and Cochrane databases for studies reporting preoperative neuroimaging analyses correlated with clinical outcomes in patients who underwent psychiatric surgery. We recorded and synthesized the methodological details, imaging results and clinical correlations from these studies.
Results: After removing duplicates, the search yielded 8388 unique articles, of which 7 met the inclusion criteria. The included articles were published between 2001 and 2018 and reported on the outcomes of 101 unique patients. Of the 6 studies that reported significant findings, all identified clusters of hypermetabolism, hyperconnectivity or increased size in the frontostriatal limbic circuitry.
Limitations: The included studies were few and highly varied, spanning 2 decades.
Conclusion: Although few studies have analyzed preoperative imaging for predictors of response to psychiatric surgery, we found consistency among the reported results: most studies implicated overactivity in the frontostriatal limbic network as being correlated with clinical response. Larger prospective studies are needed.
Introduction
Psychiatric illness affects approximately 1 in 4 people worldwide.1 Major depressive disorder (MDD) and obsessive–compulsive disorder (OCD) are among the most common and debilitating, with lifetime prevalences of 6% and 2%, respectively.2,3 Approximately 20% to 30% of MDD and OCD patients remain symptomatic and in need of novel, effective treatments despite numerous attempts at guideline-concordant optimized therapy.4,5
Various neurosurgical approaches to refractory MDD and OCD have been used for more than half a century; several procedures have experienced a resurgence of interest over the last decade.6,7 Broadly, neurosurgical procedures aiming to influence circuits that drive psychiatric disease can be categorized as lesional or modulatory; deep brain stimulation (DBS) is the most common form of the latter. Lesional procedures can be carried out as open surgical procedures involving radiofrequency (RF) thermoablation, or using less invasive techniques such as gamma-knife radiosurgery and — most recently — magnetic-resonance-guided focused ultrasound.8–10
Thanks to remarkable neuroimaging advances over the past 2 decades, neurosurgical targets and DBS programming are now informed by structural, diffusion and functional connectivity MRI, as well as by fluorodeoxyglucose positron emission tomography (18F-FDG PET) imaging.11–14 Although MDD and OCD have their own distinct neuroimaging literature, both conditions display aberrant activity throughout the limbic frontostriatal circuit.11 This circuit connects the anterior/ subcallosal cingulate and orbitofrontal cortices with the ventral striatum via the anterior limb of the internal capsule (ALIC), which then projects to the mediodorsal thalamus before looping back to the original cortical structures along the inferior thalamic peduncle.15 Stimulation and lesional targets almost all lie along this neurocircuit, including the anterior cingulate cortex, subcallosal cingulate cortex, ventral striatum and ALIC.11
As the prevalence, societal impact and neurobiology of treatment-resistant psychiatric illness becomes better understood, these surgical interventions are becoming more widely adopted. Deep brain stimulation for OCD has gained partial regulatory approval with the granting of a Humanitarian Device Exemption from the United States Food and Drug Administration after demonstrable efficacy in a randomized controlled trial and a number of case series.16–18 Many neuromodulation centres around the world now offer lesioning procedures (RF procedures, gamma-knife radiosurgery and magnetic-resonance-guided focused ultrasound) for refractory OCD and MDD.19 The results of these procedures have varied, being published largely by independent centres with small cohort sizes and varying inclusion criteria, techniques and brain targets. Despite this variation, most open-label trials consistently report 12-month response rates between 40% and 70%.20–25 In other words, nearly half of the patients who undergo these costly and invasive procedures do not make a substantial improvement, and the other half improve significantly, sometimes to the point of remission.
At present no established criteria exist to predict which patients will respond to neurosurgical treatment, nor are there established treatment-selection biomarkers to help choose between a lesional or a DBS procedure. In large psychotherapy, pharmacotherapy, electroconvulsive therapy and transcranial magnetic stimulation cohorts, neuroimaging — primarily resting-state functional MRI (fMRI) — has been used to uncover pretreatment neural connectivity patterns that distinguish likely treatment responders from nonresponders.26–30 These same neuroimaging techniques are frequently performed preoperatively on psychiatric surgery patients and likely contain valuable information for predicting response to psychiatric surgical treatments.31,32 There is a wide and varied literature looking at predictive neuroimaging in psychiatric surgery, and although a narrative review was conducted in 2011, no systematic review has been conducted to take stock of the neuroimaging prediction literature to date.33 To the best of our knowledge, this is the first systematic review to assess the existing literature of neuroimaging predictors in psychiatric surgery. Such prediction is vital to allow the implementation of personalized treatment approaches, stratified treatment based on disease subtypes and better use of increasingly limited health care resources.
Methods
This systematic review was preregistered, and details of the protocol can be found online (www.crd.york.ac.uk/prospero/display_record.php?RecordID=131151). There were no protocol deviations, and the results are reported in accordance with the a priori plan. We used the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) framework to conduct the current analysis.34 We used the Meta-analysis of Observational Studies in Epidemiology (MOOSE) criteria to evaluate and standardize the review protocol (Appendix 1, available at jpn.ca/190208-a1).35
In consultation with a health sciences librarian, we systematically searched Medline, Embase and the Cochrane Library for peer-reviewed papers from 1972 to 2019 that reported neuroimaging findings in patients undergoing psychiatric surgery (see Appendix 1 for search strategy and keywords). We chose 1972 as the start of the search to capture all possible neuroimaging studies, because the first commercially available computed tomography scanner was made available in that year.36,37
We used the following inclusion criteria: patients received DBS, capsulotomy, cingulotomy, leucotomy or subcaudate tractotomy; patients underwent preoperative imaging (MRI, PET or computed tomography); clinical outcomes were described using standardized psychiatric scales or clearly defined as “responders” and “nonresponders”; and preoperative imaging was analyzed based on its relationship to postoperative clinical outcomes. We used the following exclusion criteria: insufficient detail about how imaging analysis was carried out (we contacted authors of such papers in an attempt to determine these details); patients were operated on for nonpsychiatric indications (e.g., Parkinson disease); or research was presented in a conference proceeding or poster presentation.
Two authors (B.D. and H.S.) independently reviewed the titles and abstracts to broadly identify articles relevant to the topic. Then, using the above inclusion and exclusion criteria, the same 2 authors independently reviewed the full texts of potentially eligible studies. Disagreements were settled by consensus after discussion with a third author (N.L.). Included studies were then reviewed in detail, including appendices and supplementary data, to extract data and populate a predetermined data collection form. We converted the x, y, z coordinates of clusters of significance to the Montreal Neurological Institute (MNI) common template space using Yale’s BioImage Suite if they were reported in another template space, such as Talairach space.38
Results
Literature search
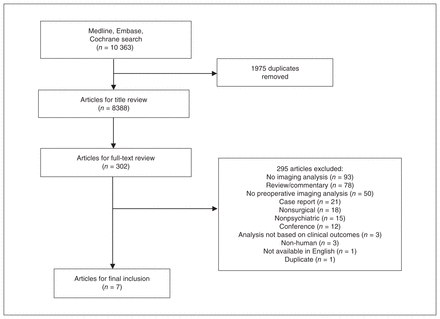
After removing duplicates, the initial search yielded 8388 articles from the 3 libraries combined. An initial title and abstract review identified 302 candidate studies. A full-text review of these papers identified 7 that met the full inclusion criteria (Fig. 1).
Flow diagram for study selection based on PRISMA. PRISMA = Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
Summary of included studies
The 7 included articles are summarized in Table 1. Articles were published between 2003 and 2018. One hundred and one unique patients were included in the analysis across the articles. The paper by Suetens and colleagues42 included the patients analyzed in Van Laere and colleagues.41 The target condition was MDD in 1 article,40 and OCD in the remaining 6 articles. The interventions used were ALIC DBS,41,42 RF cingulotomy,40,43 RF capsulotomy45 and gamma-knife radiosurgery capsulotomy.44 Although all 7 studies addressed the concept of preoperative imaging predictors of response, only those by Rauch and colleagues39 and Banks and colleagues43 were primarily aimed at identifying preoperative neuroimaging predictors of response.
Summary of included studies
Detailed description of included studies
Rauch and colleagues39 was the first published study to meet all inclusion criteria, and it paved the way for 3 of the other studies included here. In it, 11 patients with refractory OCD were treated using RF cingulotomy. Although only 2 of 11 met the prespecified responder criteria of 35%, an additional 4 of 11 improved by 25% or more. On preoperative 18F-FDG PET imaging, the authors found no voxels in the frontostriatal-thalamic circuit with significant correlations to change on the Yale–Brown Obsessive Compulsive Scale (Y-BOCS). The authors then extended their search to include regions with known structural connectivity to their initially targeted regions, identifying a cluster in the right posterior cingulate cortex (Talairach x, y, z: 16, −42, 36; 95 voxels) whose preoperative metabolism correlated with degree of Y-BOCS improvement. The authors constructed a 3 × 3 voxel region of interest centred around the peak voxel from which metabolism values were averaged, and then derived a Pearson moment correlation of r2 = 0.79 and p = 0.002. This finding remained significant at p < 0.05 when excluding the data from the greatest and poorest responders.
Dougherty and colleagues40 performed a similar analysis in 13 patients with refractory MDD treated with RF cingulotomy.40 Four of 13 exhibited a significant (> 50%) decrease in depression severity as measured by the Beck Depression Inventory. On preoperative 18F-FDG PET imaging, the authors detected 2 voxel clusters whose metabolism correlated with degree of improvement on the Beck Depression Inventory: a 24-voxel cluster in the left subcallosal cingulate (centred at MNI x, y, z −8, 24, −8) and a 231-voxel cluster in the left posterior thalamus (centred at MNI x, y, z −16, −32, 12; p < 0.001 [uncorrected]). Neither of these loci was significantly correlated with age or baseline depression severity. No r values were reported.
Van Laere and colleagues41 analyzed 18F-FDG PET scans acquired from 6 OCD patients undergoing ALIC DBS, focusing on changes between the preoperative and postoperative states. Improvement on the Y-BOCS was correlated with increased anterior subcallosal cingulate metabolism (p < 0.001 [uncorrected]); multiple comparison correction was not performed. Suetens and colleagues42 later replicated this finding with a larger cohort of 16 DBS patients that included the original 6 patients reported by Van Laere and colleagues.41 In the later study, a more liberal voxel-level threshold (p < 0.01) was used, but the result was corrected for multiple comparisons (also p < 0.01).
Banks and colleagues43 performed structural and diffusion tensor imaging analyses on data obtained from 15 patients with OCD who underwent RF cingulotomy. The authors classified patients as responders or nonresponders based on their improvement on the Y-BOCS. A voxel-based morphometry analysis, in which age and sex were included as nuisance variables, identified a grey matter cluster in the right anterior cingulate cortex that was significantly larger preoperatively in responders than in nonresponders. This cluster survived a conservative Bonferroni correction. In a diffusion tensor imaging analysis, the authors calculated the preoperative strength of connectivity between each patient’s lesion and ipsilateral brain structures as delineated in the probabilistic Harvard–Oxford structural atlas.46 To do this, each patient’s lesion masks were transformed onto their preoperative diffusion scan. Banks and colleagues43 used a previously described probabilistic diffusion tensor imaging method with a 2-fibre direction model.47,48 They determined connectivity strength based on the number of fibre tracts passing from the lesion mask and each ipsilateral atlas seed. They calculated a “laterality metric” for each lesion–seed pair, ranging from −1 to 1; negative numbers indicated stronger right-sided connectivity and positive numbers indicated stronger left-sided connectivity. Four lesion–seed connections (thalamus, putamen, pallidum, hippocampus) survived multiple comparison correction and were significantly favoured on the right in responders versus nonresponders; no lesion–seed connections were favoured on the left.
Spatola and colleagues44 reported a series of 10 patients with OCD who underwent gamma-knife radiosurgery capsulotomy. The authors reported that there were no correlations between imaging features and clinical improvement. Although this article met the literature search inclusion/exclusion criteria, the details of the neuroimaging analysis were not reported. We contacted the corresponding author, and they stated that the analysis examined changes in anisotropy with clinical outcomes, but provided no further details.
Yin and colleagues45 report a series of 27 OCD patients who underwent RF capsulotomy. Although the analysis focused on changes in functional connectivity occurring as a result of capsulotomy, a secondary analysis also looked for preoperative imaging predictors of response (as assessed by improvement in the Y-BOCS). Using standard 3.5 mm spherical basal ganglia seeds from the MDD and OCD literature, the authors identified a cluster in the dorsal anterior cingulate cortex with higher functional connectivity to the dorsal caudate in responders than in nonresponders. This degree of cingulate–dorsal caudate functional connectivity correlated with Y-BOCS improvement (R2 = 0.23), although it did not survive multiple comparison correction. Based on this seed–cluster connectivity, the authors constructed a model that was validated in an independent group of 9 patients, where it performed moderately well once an outlier was removed, although was not statistically significant (R = 0.69, p = 0.057).
Discussion
To our knowledge, this paper provides the first systematic review of preoperative neuroimaging predictors of response to psychiatric surgery. It revealed a surprising lack of studies reporting preoperative predictors; of the 302 potentially relevant articles identified from abstract screening, only 7 addressed whether preoperative imaging predicted response to psychiatric surgery. This finding was in stark contrast to the literature on antidepressants, psychotherapy and transcranial magnetic stimulation, where hundreds of neuroimaging prediction papers and recent high impact articles have been published that are now using machine-learning algorithms to make accurate out-of-sample predictions.26,27,49–51 The relatively low number of patients enrolled in psychiatric surgery trials may partly explain this shortage of predictive neuroimaging analyses. However, given the rare but serious adverse events entailed in these treatments, far more research must be devoted to using neuroimaging predictors to select patients who might benefit most and avoid unnecessary risks to patients who are unlikely to benefit.
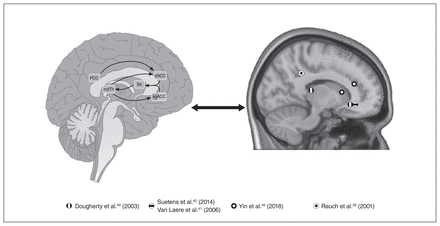
The studies identified used a variety of imaging modalities, including structural and diffusion-weighted MRI (analyzed with voxel-based morphometry and diffusion tensor imaging, respectively), PET and functional connectivity analysis, to predict outcomes after cingulotomy, capsulotomy or ALIC DBS for OCD or MDD. Despite these varied techniques, we identified a trend toward increased metabolism in frontostriatal limbic circuitry at baseline in responders compared with nonresponders (Fig. 2). Specifically, all 4 studies using employing 18FDG PET demonstrated that responders had increased FDG uptake in the cingulate (posterior, anterior or subcallosal) cortex preoperatively.39–42 Although these findings were derived from heterogeneous sources, this convergence deserves further discussion.
Mapping the findings of neuroimaging prediction studies onto known limbic neurocircuitry. Left: Sagittal schematic depicting frontostriatal-thalamic neurocircuitry implicated in psychiatric illness. Right: Spherical regions of interest centred on peak voxels of activation in Suetens and colleagues,42 Van Laere and colleagues,41 Dougherty and colleagues,40 Yin and colleagues,45 and Rauch and colleagues.39 18F-FDG = fluorodeoxyglucose; dACC = dorsal anterior cingulate cortex; mdTh = mediodorsal thalamus; PCC = posterior cingulate cortex; sgACC = subgenual anterior cingulate cortex; Str = striatum.
The cingulate cortex is increasingly viewed as a central hub for affective processing, being uniquely connected to the medial and lateral prefrontal cortices, ventral striatum, amygdala and hippocampus.14,52–54 Cortical thinning has been observed throughout the cingulate in association with depression, and the rate of this thinning is reduced with successful pharmacologic treatment.55,56 Furthermore, the subcallosal cingulate has been found to be hypermetabolic during depressive episodes, and to normalize following successful treatment with antidepressant medication.54 Hyperconnectivity of the anterior cingulate to the default mode network also correlates with severity of depressive symptoms.57 Therefore, it is not surprising that baseline dysregulation in this region is associated with response to psychiatric surgery.
In the pharmacology and psychotherapy literature, the cingulate cortex has been repeatedly identified as a predictor of response, but interestingly, hypometabolism or reduced fMRI activation has been associated with response.58–60 Cingulate hypermetabolism, which was associated with response to surgical treatments, may reflect a more treatment-resistant form of illness, and perhaps should more readily be considered for psychiatric surgery;61 patients who fail to respond to psychotherapy or medication despite having a “favourable” or downregulated cingulate may in fact have a different disease subtype and perhaps should be considered for reassessment or other treatment avenues.
Publication bias is common in neuroimaging research, and it may help explain why so few studies have been published examining neuroimaging predictors of response.62 That is, studies may have examined this question but not included findings in the manuscript because of negative results. Another related possibility is that because neuromodulation studies typically enrol a small number of patients and only a fraction of these patients are classified as “responders,” studies may lack the necessary power to examine this question. Publication of all results, significant or otherwise, such as in the case of Banks and colleagues,43 should be encouraged for future meta-analysis and assessment of publication bias.
Another potential explanation of the lack of studies on pretreatment predictors of response could be a historical overemphasis on finding “mechanisms” to explain behaviour, rather than identifying pragmatic predictive models.63,64 As articulated in an article by Kendler,65 efforts that focus only on defining etiological causation in psychiatry are deeply problematic, because psychiatric conditions involve a staggering number of etiological processes that affect different levels of behaviour. Countless papers have attempted to find a neuroimaging correlate to “explain” clinical change, although many of these could prove to merely be epiphenomena.66 Calls for more pragmatic, practical research in psychiatry have advocated for a shift toward attempts to predict, rather than explain.63
A recognized barrier to developing predictors of response to psychiatric surgery is that despite decades of neuroimaging studies, there is no “neural signature” to define common psychiatric illnesses such as MDD and OCD.67 This may be because MDD and OCD, although established clinical diagnoses, actually encompass a number of subtypes with substantial variability.26,68 Neuroimaging studies generally group patients with the same diagnosis together and fail to address heterogeneity within the disease itself.61 Conflicting or negative results may simply reflect the immense complexity of illness with signatures from different subgroups being averaged together. For instance, although “depression” is generally associated with reduced activity in the dorsolateral prefrontal cortex, some studies have described the opposite, which may simply reflect a subtype of MDD.61,69,70 A neuroimaging predictor of response in a young patient with atypical depression may be very different than in an older patient with a more symptomatic depressive phenotype; studies that pool these phenotypes may be limited in identifying robust predictive and personalized models.
Fortunately, there has been major progress toward addressing heterogeneity in psychiatry. Perhaps most notably, the Research Domain Criteria (RDoC) has been put forward by the United States National Institute of Mental Health.71 The RDoC framework was designed to guide mental health research to look beyond traditional diagnostic boundaries and consider transdiagnostic domains of functioning and constructs of behaviour with units of analysis ranging from genes or molecules to overt behavioural paradigms. Although the psychiatric surgery field lags behind, other fields are beginning to apply RDoC-based transdiagnostic approaches.72
It is of vital importance to distinguish articles that analyze pretreatment imaging in relation to clinical response, from those that analyze changes before and after treatment imaging.31,61 The former evaluate potential predictors, but the latter focus on mediators, which are unlikely to serve in treatment selection because they are measured after treatment has occurred.61 In this review, nearly 50 articles were excluded because they analyzed longitudinal neuroimaging changes over time, rather than examining preoperative imaging predictors of clinical improvement. Studies that compare pre versus post imaging, looking in particular at early post-treatment time points, could help identify nonresponders early on, allowing for redirection of their care or providing insight into disease neurobiological mechanisms.73
Future efforts at developing predictive models require increased data sharing, which may be best achieved through a consortium of neuromodulation centres following standardized imaging and treatment.31 For instance, centres with existing data from capsulotomy cases can build predictive models using variables like age, duration of illness and some imaging metrics such as functional connectivity between the dorsal caudate and subcallosal cingulate (as described by Yin and colleagues45). Ideally, a subset of data should be left out of model construction for validation; leave-one-out validation is an alternative approach for small data sets. True prediction refers to out-of-sample prediction, so these models then need to be tested and validated on other independent samples, such as data from other centres which also collect pretreatment functional imaging.74 Finally, these models should be tested prospectively on newly acquired data, with collaboration between centres to increase sample size and harmonize imaging protocols. The neuropsychiatric field in general is heading in this direction, with multiple data-sharing efforts gaining speed (such as the CranialCloud atlas for neurodegenerative disorders) but the psychiatric surgery community is still lagging behind in this regard.75,76
Limitations
This study was limited by the relatively sparse and heterogeneous data published to date, which precluded meta-analytic techniques.
Conclusion
Very few studies have investigated preoperative neuroimaging predictors of response to psychiatric surgery, despite the fact that hundreds of neuroimaging studies are available on related topics. The few studies addressing this issue point to hypermetabolism and hyperconnectivity within the frontostriatal limbic circuits, particularly involving the anterior and subcallosal cingulate cortex, as a predictor of positive response to surgery. Centres with existing preoperative neuroimaging and clinical data should work on building predictive models of outcomes and test these prospectively on future trials and across centres.
Footnotes
Competing interests: P. Giacobbe declares grants/research support: from CIHR, NARSAD, NIH and Veteran’s Affairs Canada; and honoraria and/or speaker fees from AstraZeneca, BMS, Pfizer, Eli Lilly and St. Jude Medical. No other competing interests were declared.
Contributors: B. Davidson, M. Goubran, K. Mithani, C. Pople, C. Hamani and N. Lipsman designed the study. B. Davidson, H. Suresh, and K. Mithani acquired the data, which B. Davidson, H. Suresh, M. Goubran, J. Rabin, Y. Meng, C. Pople, P. Giacobbe and N. Lipsman analyzed, B. Davidson, J. Rabin, M. Goubran and N. Lipsman wrote the article, which all authors reviewed. All authors approved the final version to be published and can certify that no other individuals not listed as authors have made substantial contributions to the paper.
- Received December 9, 2019.
- Revision received January 23, 2020.
- Accepted January 23, 2020.